(18) Transition from Sublingual to LA Injectable Buprenorphine Best Practices
Friday, April 5, 2024
9:45 AM - 1:15 PM

Phyllis Losikoff, MD
Medical Dir Research
CleanSlate Centers, Massachusetts- SC

Matthew Hanauer, MPA, PhD
Senior Data Scientist
CleanSlate Centers, North Carolina- CJ
Claudie Jimenez, MD
Chief Medical Officer
CleanSlate Centers, Tennessee - JM
Presenter(s)
Non-presenting author(s)
Background & Introduction: Extended release buprenorphine (bup) was approved by the FDA in 2017 ostensibly for persons who had been stabilized on sublingual bup. In September 2023 a second ER injectable bup was approved. Hopefully, these formulations will mitigate issues faced with sublingual films, such as diversion, misuse (Cicero et al., 2014), poor daily adherence (Tkacz, 2014), and improve treatment retention. Extended-release buprenorphine discontinuation was high in a real-world setting (Stein et al., 2022). From a programmatic standpoint, discontinuation often results in the destruction of unused injections – a costly, unintended consequence.
Programs need to adapt to emerging evidence and innovations in addiction medicine. Thus, with the availability of new formulations of ER bup injections, best practices need to be determined based on experience in real-world settings. We aimed to evaluate whether, through our experience, we can discern criteria to identify which patients are more likely to be retained on monthly ER buprenorphine for at least 4 months or more.
To assess adherence and use of non-prescribed opioids for patients who meet one or both ER buprenorphine treatment selection criteria compared to patients started on ER buprenorphine treatment who did not meet the enrollment criteria while controlling for known factors.
Methods: Eligibility criteria
Patients new to ER buprenorphine initiating monthly injections between 2021-09-01 and 2022-08-30 followed until 6.30.23 at one of 72 CleanSlate outpatient addiction treatment centers.
Outcomes:
1. Adherence: Patients remain in care and adherent to four consecutive monthly injections of ER buprenorphine.
2. Substance use: Percentage of UDS results indicating failure due to the presence of any opioid: opiates, fentanyl, methadone, and oxycodone following the first prescription of ER buprenorphine
Criteria/conditions
1. 6-weeks abstinence from opioids: The patient had no evidence of opioids in UDS testing for 6 weeks before the patient’s first ER buprenorphine injection.
2. 6-weeks in treatment: The patient has been in our treatment for at least six weeks, measured from the date of their first completed medical visit to the time of their first ER buprenorphine injection.
3. Meet both criteria 1 and 2 above.
Analyses
We used inverse probability weighting with an XGBoost logistic model to control for over 400 features (e.g., clinic location, patient demographics, diagnoses). We compared each intervention (patients in the intervention against those who were not) across both outcomes. We conducted t-tests and calculated p-values based on the adjusted averages of each outcome by intervention and control group.
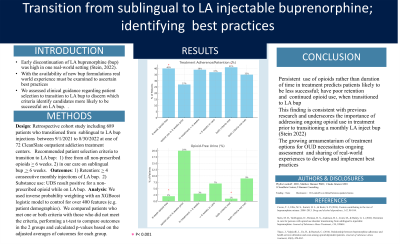
Results: 689 patients initiated ER buprenorphine from 01/09/2021 and 08/30/2022. 42% were female n = 284 and the average age was 39 (sd=10).
Patients who were opioid-free for at least 6 weeks before initiating ER buprenorphine were significantly more likely to remain on treatment for at least 4 months compared to patients who had one or more positive urines for opioids in the 6 weeks before starting ER bup injections (intervention 40% vs 27%, p <.001) and significantly more likely to abstain from opioids while on treatment (2%, control 20%, p < .001). For patients with and without 6-weeks in treatment before initiating ER buprenorphine, there was no statistically significant difference in 4 month retention (intervention 39%, control 37%, p =.472); however, those patients in the program for at least 6 weeks had significantly less opioid positive urines while on ER buprenorphine compared to patients who did not meet this criteria (3%, vs 7%, p <.001). Finally, when comparing patients who met both criteria to those who did not meet the criteria there were no statistically significant differences in adherence ( 41%, vs 35%, p=.072), while however opioid positive urine while on ER buprenorphine was significantly reduced for those who met both criteria vs patients who did not (1% vs 9%, p<.001).
Conclusion & Discussion: We found that persistent use of opioids rather than duration of time in treatment predicts poor retention for patients being retained on treatment with monthly ER buprenorphine injections. This finding is consistent with previous research and underscores the importance of addressing ongoing opioid use in OUD treatment (Stein et al., 2022). The Growing armamentarium of treatment options for OUD requires us to evaluate and share our real-world experiences, to develop best practices.
References: Cicero, T. J., Ellis, M. S., Surratt, H. L., & Kurtz, S. P. (2014). Factors contributing to the rise of buprenorphine misuse: 2008–2013. Drug and alcohol dependence, 142, 98-104.
Stein, M. D., VanNoppen, D., Herman, D. S., Anderson, B. J., Conti, M., & Bailey, G. L. (2022). Retention in care for persons with opioid use disorder transitioning from sublingual to injectable buprenorphine. Journal of Substance Abuse Treatment, 136, 108661.
Tkacz, J., Volpicelli, J., Un, H., & Ruetsch, C. (2014). Relationship between buprenorphine adherence and health service utilization and costs among opioid dependent patients. Journal of substance abuse treatment, 46(4), 456-462.
Programs need to adapt to emerging evidence and innovations in addiction medicine. Thus, with the availability of new formulations of ER bup injections, best practices need to be determined based on experience in real-world settings. We aimed to evaluate whether, through our experience, we can discern criteria to identify which patients are more likely to be retained on monthly ER buprenorphine for at least 4 months or more.
To assess adherence and use of non-prescribed opioids for patients who meet one or both ER buprenorphine treatment selection criteria compared to patients started on ER buprenorphine treatment who did not meet the enrollment criteria while controlling for known factors.
Methods: Eligibility criteria
Patients new to ER buprenorphine initiating monthly injections between 2021-09-01 and 2022-08-30 followed until 6.30.23 at one of 72 CleanSlate outpatient addiction treatment centers.
Outcomes:
1. Adherence: Patients remain in care and adherent to four consecutive monthly injections of ER buprenorphine.
2. Substance use: Percentage of UDS results indicating failure due to the presence of any opioid: opiates, fentanyl, methadone, and oxycodone following the first prescription of ER buprenorphine
Criteria/conditions
1. 6-weeks abstinence from opioids: The patient had no evidence of opioids in UDS testing for 6 weeks before the patient’s first ER buprenorphine injection.
2. 6-weeks in treatment: The patient has been in our treatment for at least six weeks, measured from the date of their first completed medical visit to the time of their first ER buprenorphine injection.
3. Meet both criteria 1 and 2 above.
Analyses
We used inverse probability weighting with an XGBoost logistic model to control for over 400 features (e.g., clinic location, patient demographics, diagnoses). We compared each intervention (patients in the intervention against those who were not) across both outcomes. We conducted t-tests and calculated p-values based on the adjusted averages of each outcome by intervention and control group.
Results: 689 patients initiated ER buprenorphine from 01/09/2021 and 08/30/2022. 42% were female n = 284 and the average age was 39 (sd=10).
Patients who were opioid-free for at least 6 weeks before initiating ER buprenorphine were significantly more likely to remain on treatment for at least 4 months compared to patients who had one or more positive urines for opioids in the 6 weeks before starting ER bup injections (intervention 40% vs 27%, p <.001) and significantly more likely to abstain from opioids while on treatment (2%, control 20%, p < .001). For patients with and without 6-weeks in treatment before initiating ER buprenorphine, there was no statistically significant difference in 4 month retention (intervention 39%, control 37%, p =.472); however, those patients in the program for at least 6 weeks had significantly less opioid positive urines while on ER buprenorphine compared to patients who did not meet this criteria (3%, vs 7%, p <.001). Finally, when comparing patients who met both criteria to those who did not meet the criteria there were no statistically significant differences in adherence ( 41%, vs 35%, p=.072), while however opioid positive urine while on ER buprenorphine was significantly reduced for those who met both criteria vs patients who did not (1% vs 9%, p<.001).
Conclusion & Discussion: We found that persistent use of opioids rather than duration of time in treatment predicts poor retention for patients being retained on treatment with monthly ER buprenorphine injections. This finding is consistent with previous research and underscores the importance of addressing ongoing opioid use in OUD treatment (Stein et al., 2022). The Growing armamentarium of treatment options for OUD requires us to evaluate and share our real-world experiences, to develop best practices.
References: Cicero, T. J., Ellis, M. S., Surratt, H. L., & Kurtz, S. P. (2014). Factors contributing to the rise of buprenorphine misuse: 2008–2013. Drug and alcohol dependence, 142, 98-104.
Stein, M. D., VanNoppen, D., Herman, D. S., Anderson, B. J., Conti, M., & Bailey, G. L. (2022). Retention in care for persons with opioid use disorder transitioning from sublingual to injectable buprenorphine. Journal of Substance Abuse Treatment, 136, 108661.
Tkacz, J., Volpicelli, J., Un, H., & Ruetsch, C. (2014). Relationship between buprenorphine adherence and health service utilization and costs among opioid dependent patients. Journal of substance abuse treatment, 46(4), 456-462.
Learning Objectives:
- List the implications for the FDA's approval of (ER) buprenorphine for opioid use disorder (OUD), focusing on how these advances could potentially mitigate issues like misuse, poor adherence, and treatment discontinuation.
- Describe the criteria that may determine better patient retention and adherence to monthly ER buprenorphine injections based on real-world evidence and machine learning approaches.
- Describe the significance of ongoing opioid use versus duration of treatment time as predictors for retention and adherence in patients on monthly ER buprenorphine treatment.
