(40) Pilot Trial of a Digital Health Intervention for Opioid Use Disorder
Friday, April 5, 2024
9:45 AM - 1:15 PM
Has Audio
- KL
Kirsten Langdon, PhD
Associate Director, Brown-Lifespan Center for Digital Health
Rhode Island Hospital/Brown School of Medicine, Rhode Island - GD
Genevieve Dash, MA
Clinical Psychology Resident
Brown School of Medicine, Rhode Island
Presenter(s)
Non-presenting author(s)
Background & Introduction: Buprenorphine-naloxone is an evidence-based treatment for Opioid Use Disorder (OUD). However, despite its efficacy, nearly half of participants discontinue treatment prematurely1,2. Novel intervention strategies that may be delivered outside of traditional treatment settings are needed to support buprenorphine uptake and maintenance. The goal of this study was to examine the preliminary efficacy of a combined web- and text message-delivered intervention (iENDURE), relative to a treatment-as-usual (TAU) control condition, in terms of improving adherence to medication, rate of illicit opioid use, experience of drug craving, and engagement/retention in treatment.
Methods: Participants were 76 individuals with OUD (38.16% female; Mage=45.91, SD=12.23) recruited from an outpatient clinic delivering buprenorphine who were randomly assigned to an intervention (iENDURE; n=56) or control (TAU; n=20) condition. The intervention condition focused on enhancing motivation for recovery and improving tolerance of distress to better manage substance use. Participants initially completed a computerized decisional balance exercise, queried about perceived barriers to treatment, and given feedback about personal motivators and recommended distress tolerance coping skills. During the second phase, participants engaged in 8 weeks of daily text messaging, which focused on promoting motivation for recovery and emphasizing adaptive strategies for distress tolerance. Participants were followed for 12 weeks and completed self-report assessments of craving, opioid use, buprenorphine adherence, and treatment engagement. Self-report measures were verified by the electronic medical record, including results of urine toxicology screens. T-tests compared mean levels of craving and engagement (percent of appointments attended throughout the study period); Fisher’s exact tests compared retention and opioid use rates across conditions; generalized estimating equations (GEEs) examined changes in craving and opioid use across condition over time.
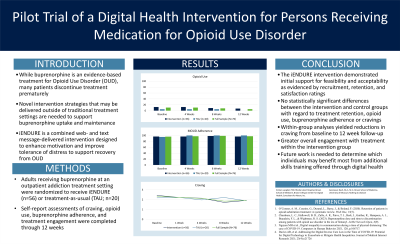
Results: Participants in the intervention group endorsed relatively high degrees of acceptability (M=20; SD=1.33 on the Client Satisfaction Questionnaire) and usability (M=77; SD=15.10 on the System Usability Scale). Additionally, intervention participants engaged with the daily text messages at a rate of 63%. At the final follow-up point (12 weeks), 81.58% (n=62) of participants were still actively in treatment. Retention rates did not differ across intervention (n=46; 82.14%) and control n=16; 80.00%) conditions (p=.53). The appointment attendance rate across the study period was 74.57% (SD=25.33); average attendance appeared slightly higher in the intervention group (77.31%; SD=23.42) than in the control group (66.63%; SD=29.45), but this difference did not reach statistical significance (t[72]=1.60, p=.11). Buprenorphine treatment adherence was uniformly high across conditions and timepoints (95.00%-100%). Across timepoints, approximately half of participants (46.05%-57.89%) denied any craving for opioids. There were no significant between-group mean differences in craving at any timepoint. In the intervention group, mean level of craving (t[55]=2.47, p=.02) was lower at 12-weeks (M=1.92, SD=1.65) compared to baseline (M=2.43, SD=2.05). At baseline, 12.50% (n=7) of the intervention condition and 5.00% (n=1) of the control condition had used opioids; at 12-week follow-up, these rates were reduced to 7.14% (n=4) and 0.00% (n=0), respectively. Rate of opioid use did not differ across groups at any timepoint (p=.28-.50). GEEs demonstrated no effects of group, time, or a group by time interaction on craving or opioid use (p=.33-.72). When covariates were included in the model, only older age predicted higher odds of denying craving for opioids (=0.07 [95% CI: 0.02-0.12]; Z=2.78, p=.005).
Conclusion & Discussion: Overall, the iENDURE intervention demonstrated initial support for feasibility and acceptability as evidenced by recruitment, retention and satisfaction ratings. There were no statistically significant differences between the intervention and control groups with regard to treatment retention/engagement, illicit opioid use, buprenorphine adherence or cravings. However, within-subjects analyses yielded reductions in cravings from baseline to the 12-week follow-up within the intervention group. Additionally, individuals in the intervention group tended to engage in more treatment appointments across the study period. In this small pilot trial results do not show an initial benefit over standard treatment, inclusive of buprenorphine and other ancillary supports (e.g., counseling, case management). Future work is needed to determine which individuals may benefit most from additional skills training offered through digital health.
References: 1. O’Connor, A. M., Cousins, G., Durand, L., Barry, J., & Boland, F. (2020). Retention of patients in opioid substitution treatment: A systematic review. PloS One, 15(5). https://doi.org/10.1371/journal.pone.0232086
2. Chambers, L. C., Hallowell, B. D., Zullo, A. R., Paiva, T. J., Berk, J., Gaither, R., Hampson, A. J., Beaudoin, F. L., & Wightman, R. S. (2023). Buprenorphine dose and time to discontinuation among patients with opioid use disorder in the era of fentanyl. JAMA Network Open, 6(9). https://doi.org/10.1001/jamanetworkopen.2023.34540
3. Nguyen MH et al., Digital inequality in communication during a time of physical distancing: The case of COVID-19. Computers in Human Behavior 2021, 120, p.106717.
4. Davies AR, et al. Addressing the Digital Inverse Care Law in the Time of COVID-19: Potential for Digital Technology to Exacerbate or Mitigate Health Inequalities. Journal of Medical Internet Research 2021; 23(4):e21726
Methods: Participants were 76 individuals with OUD (38.16% female; Mage=45.91, SD=12.23) recruited from an outpatient clinic delivering buprenorphine who were randomly assigned to an intervention (iENDURE; n=56) or control (TAU; n=20) condition. The intervention condition focused on enhancing motivation for recovery and improving tolerance of distress to better manage substance use. Participants initially completed a computerized decisional balance exercise, queried about perceived barriers to treatment, and given feedback about personal motivators and recommended distress tolerance coping skills. During the second phase, participants engaged in 8 weeks of daily text messaging, which focused on promoting motivation for recovery and emphasizing adaptive strategies for distress tolerance. Participants were followed for 12 weeks and completed self-report assessments of craving, opioid use, buprenorphine adherence, and treatment engagement. Self-report measures were verified by the electronic medical record, including results of urine toxicology screens. T-tests compared mean levels of craving and engagement (percent of appointments attended throughout the study period); Fisher’s exact tests compared retention and opioid use rates across conditions; generalized estimating equations (GEEs) examined changes in craving and opioid use across condition over time.
Results: Participants in the intervention group endorsed relatively high degrees of acceptability (M=20; SD=1.33 on the Client Satisfaction Questionnaire) and usability (M=77; SD=15.10 on the System Usability Scale). Additionally, intervention participants engaged with the daily text messages at a rate of 63%. At the final follow-up point (12 weeks), 81.58% (n=62) of participants were still actively in treatment. Retention rates did not differ across intervention (n=46; 82.14%) and control n=16; 80.00%) conditions (p=.53). The appointment attendance rate across the study period was 74.57% (SD=25.33); average attendance appeared slightly higher in the intervention group (77.31%; SD=23.42) than in the control group (66.63%; SD=29.45), but this difference did not reach statistical significance (t[72]=1.60, p=.11). Buprenorphine treatment adherence was uniformly high across conditions and timepoints (95.00%-100%). Across timepoints, approximately half of participants (46.05%-57.89%) denied any craving for opioids. There were no significant between-group mean differences in craving at any timepoint. In the intervention group, mean level of craving (t[55]=2.47, p=.02) was lower at 12-weeks (M=1.92, SD=1.65) compared to baseline (M=2.43, SD=2.05). At baseline, 12.50% (n=7) of the intervention condition and 5.00% (n=1) of the control condition had used opioids; at 12-week follow-up, these rates were reduced to 7.14% (n=4) and 0.00% (n=0), respectively. Rate of opioid use did not differ across groups at any timepoint (p=.28-.50). GEEs demonstrated no effects of group, time, or a group by time interaction on craving or opioid use (p=.33-.72). When covariates were included in the model, only older age predicted higher odds of denying craving for opioids (=0.07 [95% CI: 0.02-0.12]; Z=2.78, p=.005).
Conclusion & Discussion: Overall, the iENDURE intervention demonstrated initial support for feasibility and acceptability as evidenced by recruitment, retention and satisfaction ratings. There were no statistically significant differences between the intervention and control groups with regard to treatment retention/engagement, illicit opioid use, buprenorphine adherence or cravings. However, within-subjects analyses yielded reductions in cravings from baseline to the 12-week follow-up within the intervention group. Additionally, individuals in the intervention group tended to engage in more treatment appointments across the study period. In this small pilot trial results do not show an initial benefit over standard treatment, inclusive of buprenorphine and other ancillary supports (e.g., counseling, case management). Future work is needed to determine which individuals may benefit most from additional skills training offered through digital health.
References: 1. O’Connor, A. M., Cousins, G., Durand, L., Barry, J., & Boland, F. (2020). Retention of patients in opioid substitution treatment: A systematic review. PloS One, 15(5). https://doi.org/10.1371/journal.pone.0232086
2. Chambers, L. C., Hallowell, B. D., Zullo, A. R., Paiva, T. J., Berk, J., Gaither, R., Hampson, A. J., Beaudoin, F. L., & Wightman, R. S. (2023). Buprenorphine dose and time to discontinuation among patients with opioid use disorder in the era of fentanyl. JAMA Network Open, 6(9). https://doi.org/10.1001/jamanetworkopen.2023.34540
3. Nguyen MH et al., Digital inequality in communication during a time of physical distancing: The case of COVID-19. Computers in Human Behavior 2021, 120, p.106717.
4. Davies AR, et al. Addressing the Digital Inverse Care Law in the Time of COVID-19: Potential for Digital Technology to Exacerbate or Mitigate Health Inequalities. Journal of Medical Internet Research 2021; 23(4):e21726
Learning Objectives:
- Describe the role of technology in delivering care for Opioid Use Disorder.
- Demonstrate an understanding of factors leading to premature discontinuation of treatment.
- Describe metrics for engagement in digital health.
